
Rubius Therapeutics
Stage
IPO | IPOTotal Raised
$245MDate of IPO
7/18/2018Market Cap
0.00BStock Price
0.02About Rubius Therapeutics
Rubius Therapeutics is developing Red-Cell Therapeutics (RCTs) as a new treatment modality to address a wide array of indications including cancer, autoimmune disease, rare disease and metabolic disease. RCTs are enucleated cell-based therapies that can be engineered to have diverse activities. The company has established a platform for the rapid design, generation, testing and development of RCT products.
Missing: Rubius Therapeutics's Product Demo & Case Studies
Promote your product offering to tech buyers.
Reach 1000s of buyers who use CB Insights to identify vendors, demo products, and make purchasing decisions.
Missing: Rubius Therapeutics's Product & Differentiators
Don’t let your products get skipped. Buyers use our vendor rankings to shortlist companies and drive requests for proposals (RFPs).
Expert Collections containing Rubius Therapeutics
Expert Collections are analyst-curated lists that highlight the companies you need to know in the most important technology spaces.
Rubius Therapeutics is included in 3 Expert Collections, including Cancer.
Cancer
4,784 items
Pharmaceutical and biotechnology companies with cancer therapy drug candidates.
Regenerative Medicine
1,818 items
Regenerative medicine refers to the process of activating, replacing, engineering or regenerating human genetic material, cells, tissues or organs to restore normal function. It also includes bioengineered tissues used for in vitro testing (e.g. organ-on-a-chip, organoids).
Biopharma Tech
15,535 items
Companies involved in the research, development, and commercialization of chemically- or biologically-derived therapeutic & theranostic drugs. Excludes vitamins/supplements, CROs/clinical trial services.
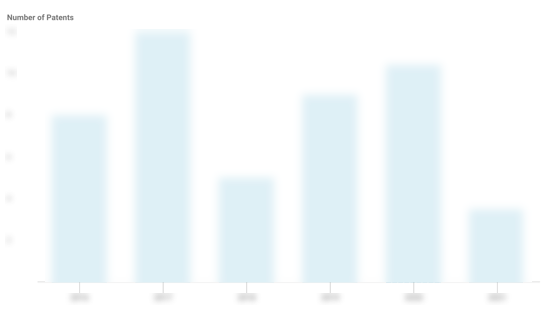
Rubius Therapeutics Patents
Rubius Therapeutics has filed 56 patents.
The 3 most popular patent topics include:
- Immunology
- Immune system
- Transcription factors
Application Date | Grant Date | Title | Related Topics | Status |
|---|---|---|---|---|
4/12/2019 | 2/14/2023 | Psychiatric diagnosis, Immunology, Immune system, Medical specialties, Clusters of differentiation | Grant |
Application Date | 4/12/2019 |
|---|---|
Grant Date | 2/14/2023 |
Title | |
Related Topics | Psychiatric diagnosis, Immunology, Immune system, Medical specialties, Clusters of differentiation |
Status | Grant |
Latest Rubius Therapeutics News
Apr 20, 2023
Billion by 2030 | CAGR of 6.2% The Global Phenylketonuria Market Size was valued at USD 610.3 Million in 2022 and the worldwide phenylketonuria market size is expected to reach USD 1121.1 Million by 2032, according to a research report published by Spherical Insights & Consulting. Companies Covered: Daiichi Sankyo Company Limited, Cambrooke Therapeutics, American Gene Technologies International, Inc., Dimension Therapeutics, Inc., Synthetic Biologics, Inc., BioMarin Pharmaceutical, Inc., Erytech Pharma SA, Danone Nutricia, Censa Pharmaceuticals, Homology Medicines, Inc., SOM Innovation Biotech SL, Mayo Foundation for Medical Education and Research, Rubius Therapeutics, Inc. and among others. April 20, 2023 07:45 ET Pune, INDIA New York, United States , April 20, 2023 (GLOBE NEWSWIRE) -- The Global Phenylketonuria Market Size is to grow from USD 610.3 million in 2022 to USD 1121.01 million by 2032, at a Compound Annual Growth Rate (CAGR) of 6.2% during the forecast period. Get a Sample PDF Brochure: https://www.sphericalinsights.com/request-sample/1704 The body overproduces the amino acid phenylalanine due to a rare hereditary condition called phenylketonuria. All proteins and certain artificial sweeteners include the amino acid phenylalanine. The non-essential amino acid tyrosine is produced by the body's phenylalanine hydroxylase enzyme. Dopamine, norepinephrine, and epinephrine are just a few of the neurotransmitters that the body requires tyrosine to produce. PKU can be brought on by a mutation in the PAH gene, which encodes the phenylalanine hydroxylase enzyme. Diagnostic procedures, treatments, and dietary supplements for PKU sufferers are all part of the global phenylketonuria market. The increasing awareness of phenylketonuria among medical professionals and the general public is one of the key factors anticipated to propel the growth of the phenylketonuria market during the forecast period. Also, governments and regulatory organizations are supporting and subsidizing the development of novel phenylketonuria treatments, which is fostering innovation and introducing novel products to market. There have been substantial improvements made in the creation of new PKU drugs, including gene therapy, pharmacological therapies, and enzyme replacement therapy. Furthermore, phenylketonuria treatments, such as gene therapy, pharmacological therapies, and enzyme replacement therapy, can be extremely expensive. For some patients, particularly those in low- and middle-income nations, this might hinder their access to these types of drugs. Due to the frequency of this disorder and the limited availability of specialized medical meals, conducting clinical trials for novel PKU treatments can be challenging. This may reduce the number of new therapies that are available and delay their development. Browse key industry insights spread across 200 pages with 130 market data tables and figures & charts from the report on the " Global Phenylketonuria Market Size , Share, and COVID-19 Impact Analysis, By Product Type (Drugs, Nutrition Supplements, Others), By Type (Hyperphenylalaninemia, Mild PKU, Moderate and Variant, Classic PKU), By End Users (Hospital Pharmacies, Online Pharmacies, Drug Stores, Pediatric Clinics, & Others), By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2022– 2032." Get Detailed Report Description Here: Buy Now Full Report: https://www.sphericalinsights.com/checkout/1704 COVID 19 Impact The COVID-19 pandemic has had a significant impact on the global healthcare industry, including the phenylketonuria (PKU) market. The pandemic has disrupted healthcare services around the world, and many individuals with PKU may experience delayed diagnosis or treatment as a result. This could lead to long-term consequences for individuals with PKU and could impact the growth of the phenylketonuria market. The economic impact of the pandemic could make it more difficult for some patients with phenylketonuria to afford the specialized medical foods and nutritional supplements required to manage their condition. This could impact treatment adherence and could impact the growth of the PKU market. The pandemic has accelerated the adoption of telemedicine and remote healthcare services, which could provide opportunities for individuals with PKU to receive care and support from home. This could improve access to care and support for individuals with PKU and could potentially benefit the growth of the PKU market. The nutrition supplements segment is dominating the market with the largest market share over the forecast period. Based on the product type, the Global Phenylketonuria Market is segmented into drugs, nutrition supplements, and others. Among these, nutrition supplements dominated the market over the forecast period due to Specialized medical foods and nutritional supplements developed to deliver the required nutrients while reducing phenylalanine intake are included in the nutrition supplements industry. These goods might comprise infant formulae, beverages, and snack bars that are high in protein, vitamins, and minerals but low in phenylalanine, which could dominate the market share over the forecast period. The hospital pharmacies segment is influencing the market over the forecast period. Based on the end users, the Global Phenylketonuria Market is divided into hospital pharmacies, online pharmacies, drug stores, pediatric clinics, & others, the hospital pharmacies segment held the largest market share owing to the hospital infrastructure can hold a significant number of patients. In order to prevent any potential mistakes, most people also prefer to get their medications from hospital pharmacies and then have them reviewed by a doctor. Inquire Before Buying This Research Report: https://www.sphericalinsights.com/inquiry-before-buying/1704 North America is dominating the market with the largest market share over the forecast period. North America is predicted to expand at the highest CAGR over the projection period due to the increased awareness of PKU and other rare diseases among patients and healthcare professionals in this area, North America is predicted to expand at the highest CAGR over the projection period. Government programs to raise awareness of these illnesses are also promoting market expansion in this region. Asia Pacific is anticipated to have significant growth over the forecast period due to the increase in the older population in countries like China and India. The expansion of medical facilities and the localization of generic drug manufacturers are primarily responsible for the increase in the region's revenue. Competitive Analysis: The report offers the appropriate analysis of the key organizations/companies involved within the global market along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market. Major vendors in the Global Phenylketonuria Market include American Gene Technologies International, Censa Pharmaceuticals, Daiichi Sankyo Company Limited, Cambrooke Therapeutics, Inc., Dimension Therapeutics, Inc., Synthetic Biologics, Inc., BioMarin Pharmaceutical, Inc., Erytech Pharma SA, Danone Nutricia, Homology Medicines, Inc, SOM Innovation Biotech SL, Mayo Foundation for Medical Education and Research, Rubius Therapeutics, Inc., and Other key players. Get Discount At @ https://www.sphericalinsights.com/request-discount/1704 Recent Development In July 2022, Preclinical evidence for Jnana Therapeutics' flagship program, which could be the first oral strategy of its kind to treat PKU, has been reported. At the Vancouver, Washington, 2022 National PKU Alliance Conference, data has been presented. Browse Related Reports Global Breast Cancer Market Size, Share, and COVID-19 Impact Analysis, By Cancer Type (Hormone Receptor, HER2+), By Therapy (Targeted Therapy, Hormonal Therapy, Surgery & Radiation Therapy, Biologic Therapy), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, & Others), By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2022 – 2032
Rubius Therapeutics Frequently Asked Questions (FAQ)
Where is Rubius Therapeutics's headquarters?
Rubius Therapeutics's headquarters is located at 399 Binney St, Cambridge.
What is Rubius Therapeutics's latest funding round?
Rubius Therapeutics's latest funding round is IPO.
How much did Rubius Therapeutics raise?
Rubius Therapeutics raised a total of $245M.
Who are the investors of Rubius Therapeutics?
Investors of Rubius Therapeutics include Flagship Pioneering.
Who are Rubius Therapeutics's competitors?
Competitors of Rubius Therapeutics include TetraGenetics.
Compare Rubius Therapeutics to Competitors
selectION is developing a class of medicines to treat autoimmune diseases and other ion channel related disorders.
Calypso Biotech is a biopharmaceutical company that develops monoclonal antibodies for the treatment of immune pathologies with unmet medical needs.

Metrion Biosciences is a UK-based Contract Research Organization (CRO) that focuses on delivering a range of ion channel drug discovery services. The company provides electrophysiology screening services to support client medicinal chemistry optimization programs, CiPA compliant cardiac safety profiling assays, neuroscience assays and translational assays.

ROME Therapeutics is developing therapies for cancer and autoimmune diseases by harnessing the power of the repeatome - vast stretches of uncharted genetic material that have long been dismissed as "junk DNA". With several drug targets identified and multiple discovery programs underway, ROME is moving to leverage this new frontier in biology. To lead this exploration, ROME has assembled a team of leaders across fields including oncology, immunology, virology, and machine learning.

Kineta (NASDAQ: KA) is a biotechnology company that focuses on developing therapeutics in areas of autoimmune disease, viral disease, and chronic pain. The company was founded in 2008 and is based in Seattle, Washington.

Cue Biopharma engineers with disease-relevant T-cells for the treatment of cancer and autoimmune disease. Its technology platform delivers amplifying or attenuating signals to specific T-cell subsets and address any T-cell-mediated disease. The company was founded in 2014 and is based in Boston, Massachusetts.
Discover the right solution for your team
The CB Insights tech market intelligence platform analyzes millions of data points on vendors, products, partnerships, and patents to help your team find their next technology solution.